End of summer, with the cooler night time temperatures, prompts area residents to close their pools for the year. Where does all that pool water go?
In my neighborhood, almost half of the neighbors have pools, most are in-ground pools and a couple are above-ground pools. The pool water is gorgeous- clear, blue, inviting for a cool dip on a hot summer’s day. I often have pool envy, especially when it’s so hot that I wonder if I could fry an egg on the sidewalk. According to Mainetti and Mainetti, LLC, there are about 17,000 private pools in Dutchess County, NY, which is about 60 miles north of NYC.

There’s a lot of chemistry in maintaining a safe and gorgeous pool. Most residential pools keep their pH balanced and also prevent the growth of algae or harmful bacteria by adding chlorine to the water. Let’s take a closer look since we ingest that water when we go swimming, of course immerse ourselves in it and then….where does that chemically treated water go when the pool is closed?
The Basics of Pool Chemistry
To keep a pool clear and sanitized, the pH needs to be near our own body’s pH, in the range of 7.2 – 7.8. (That’s a huge range- review the earlier newsletter on pH.) If you live in an area with “hard water,” the calcium levels need to be around 300 ppm (parts per million). It’s also important to maintain the chlorine levels at around 2 ppm and filter out things like various solids.
The tools for keeping the pool clear and balanced are chemicals that you add, ways to test the water chemistry, a filtering water pumping system to circulate the water, a pool vacuum for the things that settle to the bottom and a net for scooping up large things, like leaves or, gross, dead frogs or other things that might fall in and die.
A big issue for swimming pools is algae growth. The chlorine levels (at the proper pH levels) keep algae from growing, but you have to keep the chlorine and pH levels balanced and the water moving and filtering, to prevent the algae from growing. Often folks discover algae growing and have to add an “algicide” to the water. Yes, algicides are a kind of pesticide.
The chemistry of opening a pool
From September til May, the pools have been covered with a black tarp that’s tacked down all around the pool. Usually, the pools have been drained to below the level of the filtration system. Depending on the pool size this might be 1000 gallons. [This is then perhaps 170,000 gallons in Dutchess County alone.]
All sorts of weather has happened throughout the closed pool months: rain, snow, ice. Leaves have fallen. Temperatures have gone up and down. The water chemistry definitely gets out of whack. Residential pools open in the spring, typically in our area sometime in May. Most of the time, homeowners go through a ritual of chemical treatments to rebalance the water, adding pool shock chemicals, chemicals to make the water alkaline, then chemicals to balance the pH, chlorine to reestablish the right level of chlorine to keep the pool from harboring algae or bacteria, and chemicals to stabilize calcium levels. And, of course, water needs to be added too. Perhaps 1000 gallons, which some homeowners simply get from their house supply of potable drinking water.
What are the chemicals?
Alkalinizer: this is sodium bicarbonate (which is baking soda) and adds some base to the water to help with keeping more of a pH balance. Back in an earlier newsletter, we learned about pH and how pure water is pH 7.0, which is the value of how much water (H2O) has dissociated into H+ and OH–. A buffer system, of salts in the water, can help keep a water solution in balance, by absorbing H+ if that gets too high. For the swimming pool, most of the challenges to maintaining pH will tend to shift it acidic. Adding sodium bicarbonate to the water gives it a buffer capacity to absorb some of those challenges. If the pH gets too basic, you can add sodium bisulfate which is an acid, to bring it down.
Pool shock: calcium hypochlorite, also known as bleaching powder, or sodium dichloro-s-triazenetrione (dichlor)

Pool shocking involves working with high concentrations of agents that can be very toxic even to people. Chlorine gas is toxic, damaging lungs and eyes. Chlorine is also harmful to skin. So, you’re supposed to wear gloves, long pants, long sleeves, eye protection and preferably a respirator style mask. (Have you seen pool folks do this when they open their pools? Nah, shorts, flip flops – that’s the pool opener’s typical garb) After you shock a pool, you’re supposed to stay out of it for at least 24 hours…..in my experience with municipal pools, the post-shock wait time is rarely a full 24 hr. Kids can’t wait to jump back into the pool after someone’s had an “accident” and the pool was shocked.
A chlorine stabilizer chemical,
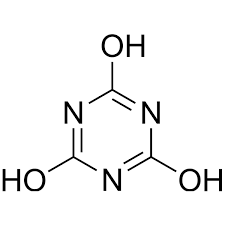
cyanuric acid, is often added to help keep the chlorine in a “free” form so that it continues to be toxic to algae and bacteria. Chemicals in pool water, including urine, sweat and lotion residues, can convert chlorine to a “combined” form, called chloramines. These can be corrosive to pool equipment, and very irritating to eyes and your nasal and lung passages. There was a time when the pool I regularly swim in had a high level of chloramines and, when I shampooed my hair after swimming, blobs of my hair came out of my scalp! Yikes! Chloramines, which kill fish, frogs and other aquatic creatures, are less effective at preventing algae growth. Shocking the pool can break up the chloramine compounds. (Chloramines are one big reason public pools ask you to shower before swimming, to rinse off sweat and lotion residues that interact with free chlorine to form chloramines.)
To keep the chlorine and pH levels balanced, you also need to have the right amount of calcium. Too much calcium and too little calcium affect both the pH and the free chlorine and can also corrode the pool itself. Calcium chloride can increase the calcium (and so increase the hardness of the water). This stuff can also be used as an ice melt!

If your calcium levels get too high and the water is cloudy, you might have to empty some water and add new. You can manipulate the pH, making it more acidic to dissolve some calcium, but this is a tough problem to manage. Best to avoid in the first place.
The temperature of the water can also affect all these chemical levels. For example, if there’s a stretch of really chilly mornings after a pool is opened, the ability of calcium, chloride and other chemicals to stay dissolved in water changes, which can affect pH and other aspects of pool water balance.
Wow! That’s a lot of chemistry! Back to the water…..where does it go when the pool is closed for the fall/winter?

When people close their pools for the winter, they drain many gallons of water that has held levels of chemicals that are toxic to algae, bacteria, and aquatic wildlife. Just releasing that water into the environment can be toxic to plants and soil and water organisms. You’re supposed to empty that pool water into your municipal water system so that it goes through the same water treatment as the waste water from your house. But, if you have a septic system, that can overwhelm the system and can also disrupt the biology of the septic field, which relies on bacteria to work on your human waste.
If folks don’t have their home waste water connected to a municipal sewer system, they are supposed to empty their pool water on their lawns. To protect the soil and landscaping in your yard, the pool owner can stop adding chlorine for a couple of weeks, when sunlight and evaporation can lower pool chlorine levels and then drain the water into the lawn. You have to be careful that the pool levels of chlorine really are low, and a lot of folks are in a bit of a hurry to close the pool and might take short cuts. In my experience, most of the neighbors, especially the ones who also own in-ground pools, also really care about having a green, perfect, monoculture lawn. So, the temptation is to empty the water into the street, letting the storm drain system take the water. Which, of course, evades water treatment and ends up directly in the Hudson river and other water sources in the area. It’s not legal, but who monitors?
Another question: might the gallons of dumped pool water seep into area drinking water wells, contributing to chlorine levels? All that pool water dumped on your lawn will go downhill to neighbor yards if you have any slope and also percolate into the soil, moving towards lower lying areas. Not so environmentally friendly, is it?
I LOVE pools. I think that people need to better understand the ways in which this incredible luxury might be harmful to others, especially non-humans, and take steps to minimize that harm. Even installing a water filter (like you put on your kitchen sink), would filter out chlorine and other pool chemicals and make the draining of pools safer for everyone.
This newsletter is devoted to explaining how biology works and how it is relevant to our daily lives. Most of us stopped learning about biology in high school or even middle school. And the way we learned it was as isolated concepts and vocabulary to memorize. I hope that this newsletter helps you rekindle that love of biology and might even help with better understanding of some of the important biology all around us. Please share this with anyone you think might want to take a look.
Thanks for reading!





