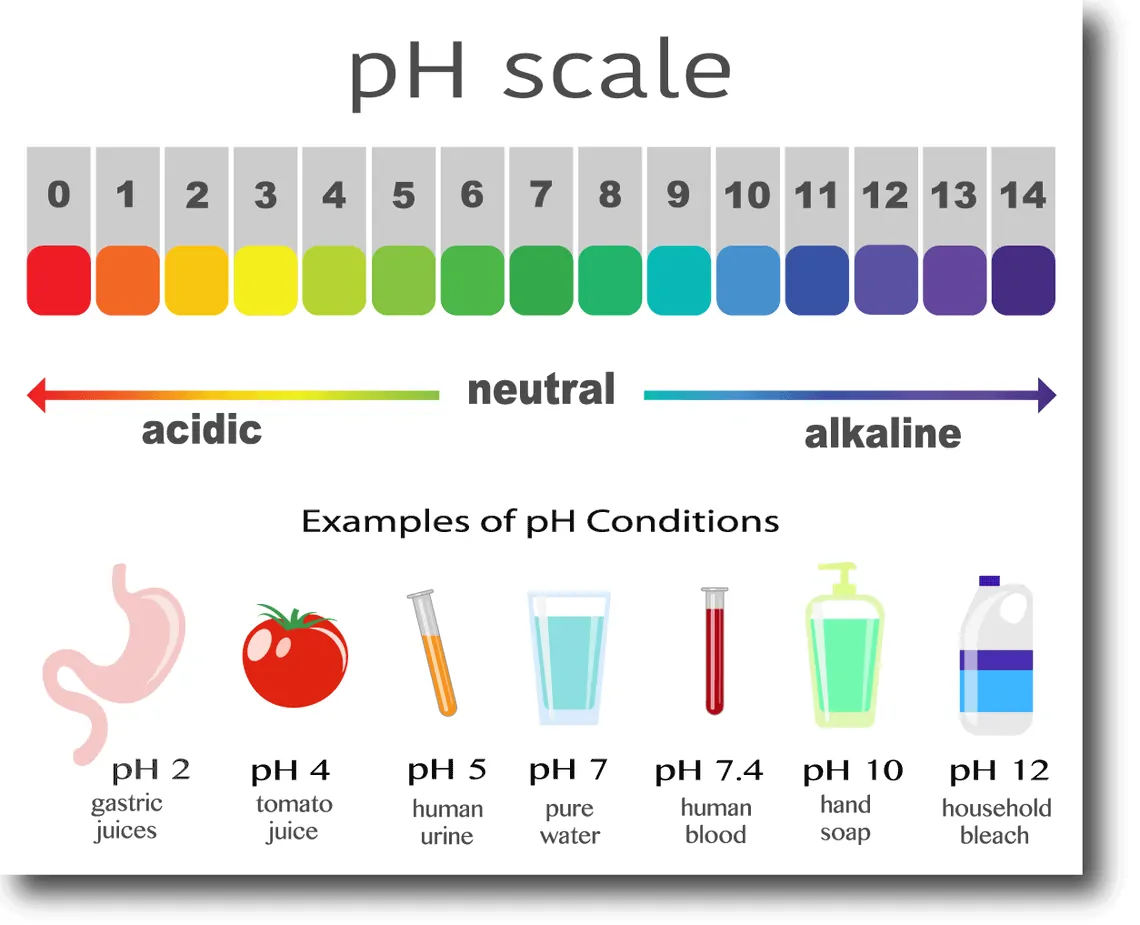
featured image courtesy of: https://www.allaboutsmilesde.com/what-is-oral-ph-and-how-does-it-affect-your-health/
If you’ve ever watched Breaking Bad, you remember how a murdered body was disposed of by dissolving it, even the bones, in a bathtub full of hydrofluoric acid (the bathtub itself was corroded/dissolved by the acid). Of course, MythBusters and others pointed out that this fictional, but nonetheless thrilling idea, might not fully dissolve human bones, but certainly makes the remains unrecognizable. A recent article published in Forensic Science International took on a similar grisly experiment and showed hydrochloric acid was pretty darned destructive to human bone in as little as 4 hr. I think hydrochloric acid (HCl) would have been a better choice for an actual disposition of a murdered corpse than hydrofluoric acid (HF) because HF also eats through glass and metals, but I’m sure it was chosen for the episode because of the bathtub disintegration. How does acid work?
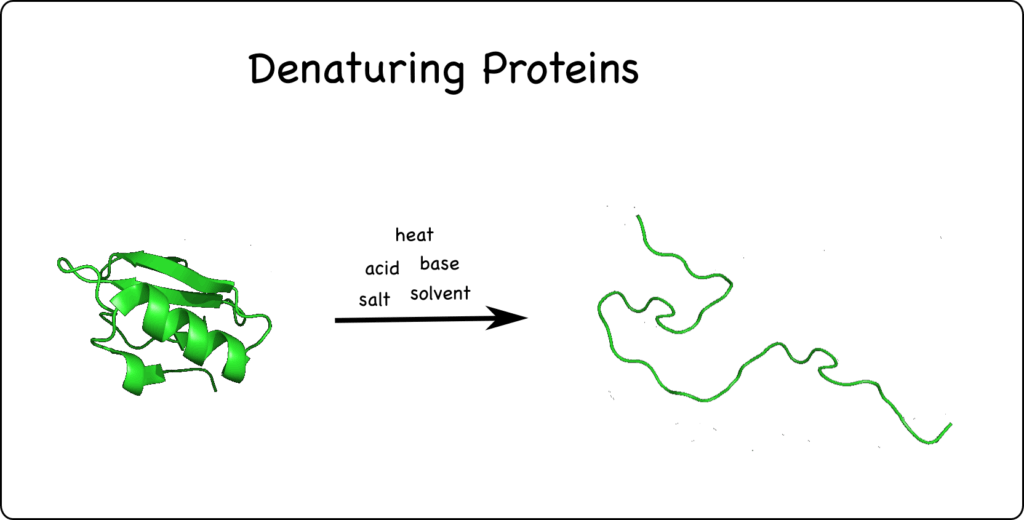
In the last post, I talked about how polar chemicals in water solution dissolve by interacting with water molecules and that some of these release H+ into the water, making the H+ concentration higher, which is the definition of acidic. The more acidic, the more reactive. Those H+ are super reactive and can interfere with the shape of proteins or even break them apart, which means they can no longer function. The term for this protein damage is denaturation. Proteins in our skin’s epidermis that are exposed to acid leads to acid burns as the proteins are destroyed. Or, the tender membranes of our nasal passages if we breathe in acid vapors get damaged as the proteins are destroyed. All of our body parts are what they are and do what they do courtesy of the many types of proteins they are made up of. Acids are bad news for most proteins. It’s bad for other body chemicals, too.
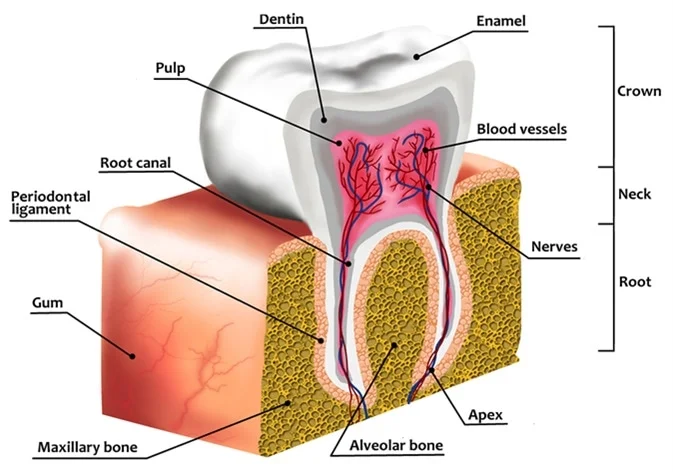
The proteins that make up our teeth and bones are really tough. Teeth proteins like dentin and enamelin cluster together with calcium-based minerals to form a super durable and tough outer layer of enamel. Normally our saliva helps to keep the pH in our mouth fairly neutral, even if we are eating foods that are acidic (like oranges or hot sauce) or drinking acidic soft drinks, coffee or tea.
Cola (even diet versions) has phosphoric acid in it bringing it to a pH of 2.5, which is a strong acid that can weaken the enamel structure and make your teeth more susceptible to the bacteria that can cause tooth decay. If you put teeth into a cup filled with cola and leave them there, the damage from the phosphoric acid can be pretty severe. Here’s a great video of what happens after a week in the soda. When I was in grad school, I got on a kick of drinking diet Coke every day. When I went to the dentist later that year, I had seven cavities! Before that, I had only had a couple over more than 20 years. Needless to say, I don’t drink soda anymore. I drink coffee in the morning, but brush my teeth right after to get rid of acid residue. I’ve hardly had any cavities in the subsequent decades.
I do drink seltzer, which has a pH of about 5. It’s an acid, but not as strong as cola. My saliva can help to neutralize the acid (from the dissolved carbon dioxide that gives seltzer it’s fizz), so I’m not too worried about it making my teeth decay. But, like the ocean acidification I talked about last week, if I soaked my teeth in seltzer for a long time, the enamel would likely get thinner, like a seashell thins in an acidified ocean.
Here’s a great article about how pH can weaken the calcium-crystal structure of enamel and how fluoride strengthens enamel to resist acid damage: https://pmc.ncbi.nlm.nih.gov/articles/PMC6151498/
Ok, back to pH and our bodies.
Our bodies are incredible- truly. We eat food and drink various substances and our GI tract digests it into the building blocks that build our bodies. We can think of our bodies as coordinated and collaborating compartments. Our digestive system is basically a long compartmentalized tube. Here’s a picture from the textbook I use in my Human Physiology class. Look at it closely for a few seconds. Notice that the GI system is organized into different regions in an overall self-contained compartment that is open to the outside world at the mouth and the anus, but is separated from the rest of the body compartments (blood, bones, skin, etc).
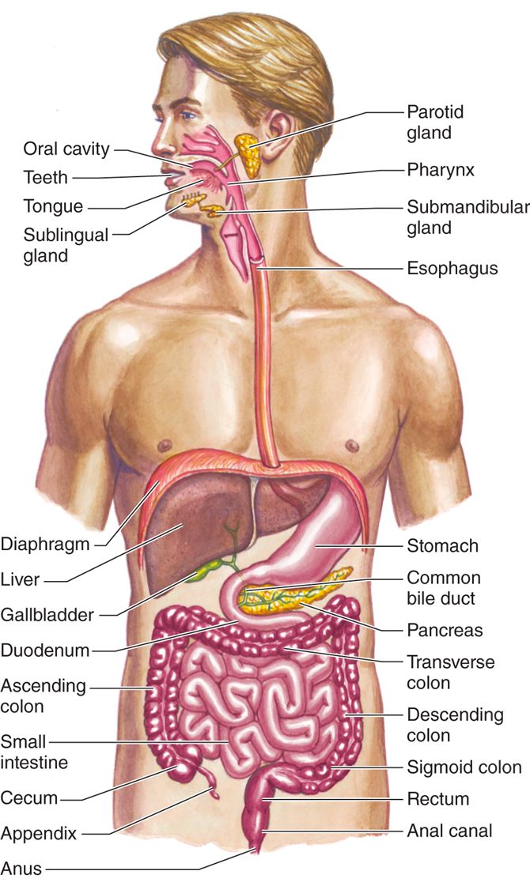
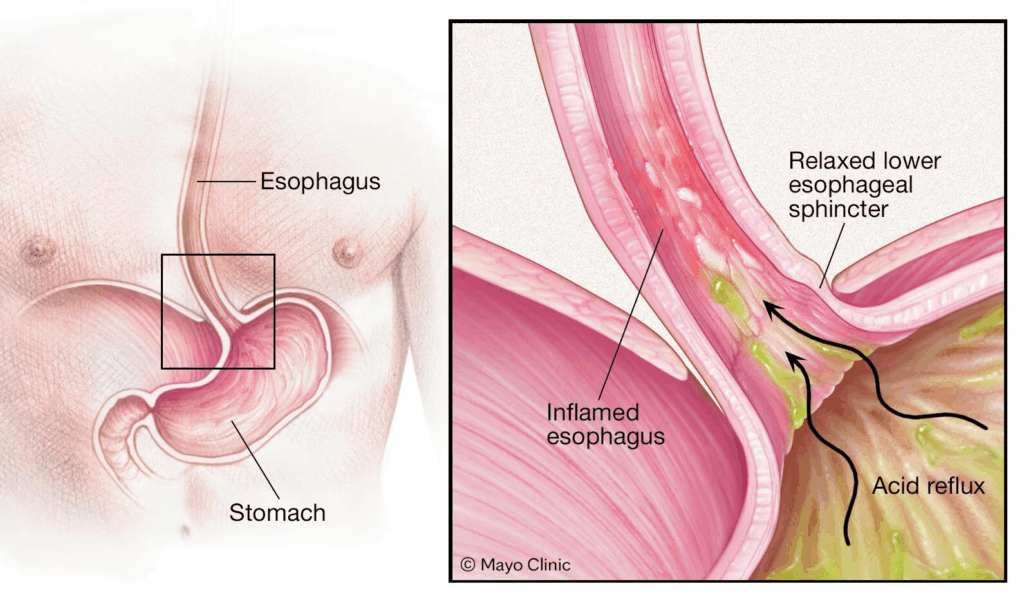
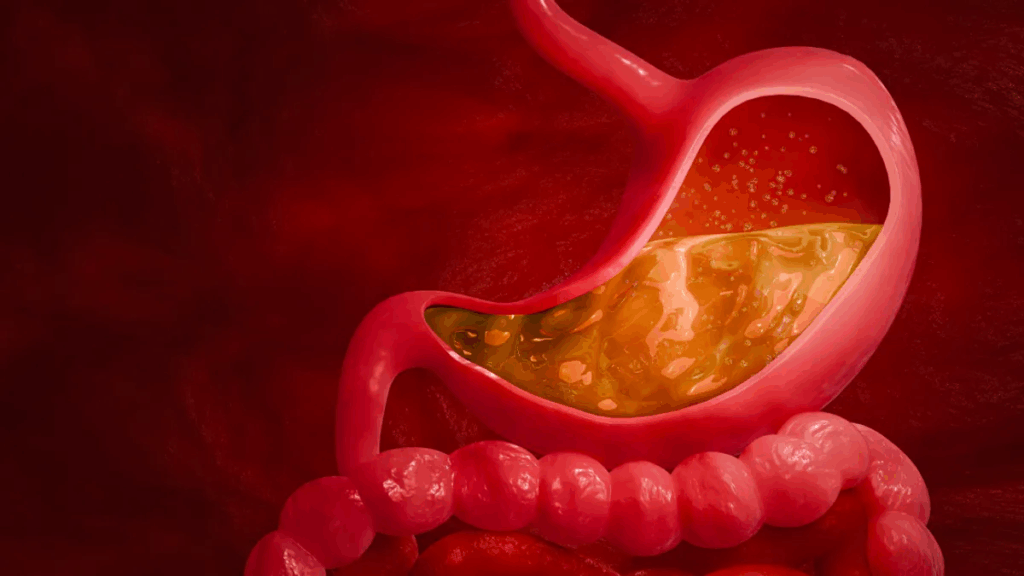
We chew our food and mash it up, mixing it with our water-based saliva. Then, we swallow and the paste travels down our esophagus, which is about 10 inches long, to the stomach. The stomach starts oozing cups of concentrated hydrochloric acid (about 8 cups a day) and churning to mix it all together. The stomach contents’ pH drops to about 2. This wicked acid level is crucial for protecting us from many of the contaminants in our food, like plant toxins or bacteria. There’s an enzyme made by stomach cells, pepsin, that needs this acid environment to break apart proteins. The stomach acid is confined to the stomach and the stomach lining itself is coated with a super thick layer of mucus, to protect it from the acid. Some people have a weakness in the ring of muscle that separates the esophagus from the stomach and, when the stomach really gets churning, some of the acid-rich material spurts backwards, causing a heartburn condition called GERD (gastroesophageal reflux disease), or acid reflux.
You can also get occasional heartburn (which isn’t your heart burning- it’s the burning acid sensation in the esophagus, which isn’t protected with thick mucus like the stomach lining). That kind of heartburn can happen if you have a really distended stomach from overeating (think Thanksgiving dinner!), or you eat a lot and are lying down (like lounging on the couch watching the Superbowl of the finale of Love Island eating nachos). Gravity helps keep the food moving through the stomach and into the intestines. That’s one reason taking a slow walk or even just standing up after dinner can be helpful. My Uncle Tommy used to stand up at the Thanksgiving table to let his stomach “settle” so he could cram in the pumpkin pie. My other uncles and my dad would roll into the living room and collapse on the sofa like beached whales (and then fall asleep in front of the football game). It’s notable that my Uncle Tommy was able to join us kids outside for a game of hide and seek. Another fun factoid about our digestion is the time it takes for a meal to be fully processed: it takes about two hours for the food paste to make it out of the stomach; about 3-6 hours through the small intestine and up to 72 hours through the colon.
From our stomachs, our acid paste-like food is squeezed in small amounts from the stomach into the small intestine, where about 8 cups (half a gallon!) a day of alkaline watery juice is released by the pancreas to neutralize the acid so that our digestive enzymes can get to work doing the chemical breakdown of our food into our body’s building blocks of blood sugar, amino acids and more. These nutritional chemical building blocks are absorbed from the small intestine into the blood, pass through the liver to remove certain toxins like alcohol, and make their way to the organs and tissues that are constantly renewing and refreshing and doing the work of the body. What doesn’t get absorbed in the small intestine enters the colon (large intestine), where our own personal horde of bacteria, called our microbiome, feast off what’s left, giving us more nutritional chemicals, like Vitamin K and Vitamin B-12. The colon also is where a lot of the water from all that amazing intestinal chemistry gets absorbed back into our body. In the end, we poop out the waste.
The crazy acid pH in the stomach surges every time we eat. But that compartment is kept separate from the other compartments in our body, which are close to neutral pH, near 7. When you are healthy and just eating typical foods in typical amounts, the acid produced by your stomach and the neutralization in your small intestine do not affect the pH of the blood or other parts of our body. This means that the ideas of having an “alkaline diet” to alter body pH are really not changing blood or body pH.
The most common situations where your guts affect your blood pH are when you have excessive vomiting where you lose substantial stomach acid that isn’t replaced, which can put your body in a state of alkalosis (basic pH), or when you have diarrhea, where you can quickly lose lots of the watery and alkaline solution that came from the pancreas, making your body more acidic (called acidosis).
What keeps our blood and body in a tight pH balance is an amazing collaboration between our lungs and our kidneys. That’s where some really cool acid/base pH magic happens. We’ll get into this next time.
Coming up in the next post:
Crucial to our health is maintaining a tight pH balance in our blood and bodies of 7.35. Next time, I’ll talk a little bit about how our lungs and kidneys work together to keep us balanced. Hope you’ll tune in.
This newsletter is devoted to explaining how biology works and how it is relevant to our daily lives. Most of us stopped learning about biology in high school or even middle school. And the way we learned it was as isolated concepts and vocabulary to memorize. I hope that this newsletter helps you rekindle that love of biology and might even help with better understanding of some of the important biology all around us. Please share this with anyone you think might want to take a look.
Thanks for reading!