You’ve probably heard that one big concern about the growing amount of carbon dioxide in the air, from humanity’s burning of fossil fuels, is ocean acidification. I noticed that most internet sources describe how oceans get acidified, but they don’t really explain what that means- especially what pH is.
pH is not one of those ideas that most of us would list in our daily thoughts. Acid-Base Balance. What’s that even mean and why should we care? Honestly, most of us don’t. We encounter the concept of pH in middle school science and again in high school chemistry or maybe biology. Most of the time, it’s just explained like this, from the EPA website:

“The pH scale measures how acidic or alkaline a substance is. The scale ranges from 0 to 14. A pH of 7 is neutral. A pH less than 7 is acidic, and a pH greater than 7 is basic. Pure water is neutral, with a pH of 7.0. When chemicals are mixed with water, the mixture can become some level of either acidic or alkaline. Vinegar and lemon juice are acidic substances, while laundry detergents and ammonia are alkaline.”
https://www.epa.gov/goldkingmine/what-ph
Okay, this definition seems like some arbitrary feature of chemicals and that, if we’re told it’s important to be balanced (somewhere near 7?) then….ok….guess we gotta be. I love in that picture above where 4.2 is located it says “All Fish Die.” Dramatic.
But yes, dramatic! Look at the picture again, where it shows the measured pH of acid rain. When I was a kid there was a lot of public worry about acid rain. I used to think that the rain would be so acidic that it would melt my umbrella and my skin. It is pretty scary to others even today. A 2023 movie centered on a toxic rain storm that was so acidic that it killed people and melted suspension bridges.
The EPA explanation of pH and what’s an acid and what’s a base (or is alkaline) really doesn’t tell us what pH is and why being out of balance is bad. Why is acid rain like in the movie so scary?
pH, acids, bases, are all related to that amazing property of water, polarity. In an earlier post, I mentioned that water’s polarity means it can dissolve a lot of stuff- by basically pulling molecules apart to a degree and surrounding them with water molecules. Water molecules pull on each other, too, leading to some of them in the form of H+ and OH– (called ions). In a solution of pure water, there’s a balance of mostly water molecules (engaging in those bond interactions I discussed in an earlier post) and these ions. This balanced state is called neutral, or pH 7. [If you want to know how the pH numbers arise, here’s a great site.]
Things that dissolve in water also have polarity. Parts of those chemicals, particularly H+ and OH–, can be pushed and pulled by water’s polarity and even come off those chemicals. This is called dissociation and is one process involved in dissolving. The dissociation of H+ and OH– into the water makes the chemicals more reactive with other chemicals. It makes the water more reactive, too. Some chemicals are more likely to lose H+ into water, which will make the water have a higher percentage of H+, making it more acidic. Other chemicals give up OH– or grab H+ from the water, making it (the water and chemical solution) more basic. Both acid solutions and basic solutions are more reactive with other chemicals. It is that reactivity that is important in how acids and bases work.
Where does acid rain come from?
When cars, trucks, boats and factories burn fossil fuels, they release chemicals into the air. Chemicals like carbon dioxide, of course, but also sulfur dioxide (SO2) and nitrous oxide (NO2). These chemicals interact with the water in the atmosphere (especially in clouds)- just like they would in a lake or other body of water. As these polar molecules interact, they dissociate and combine together, forming sulfuric acid and nitric acid, which then rain down from the sky. The acid rain or acid sky dust lands in water and land, making both more acidic.
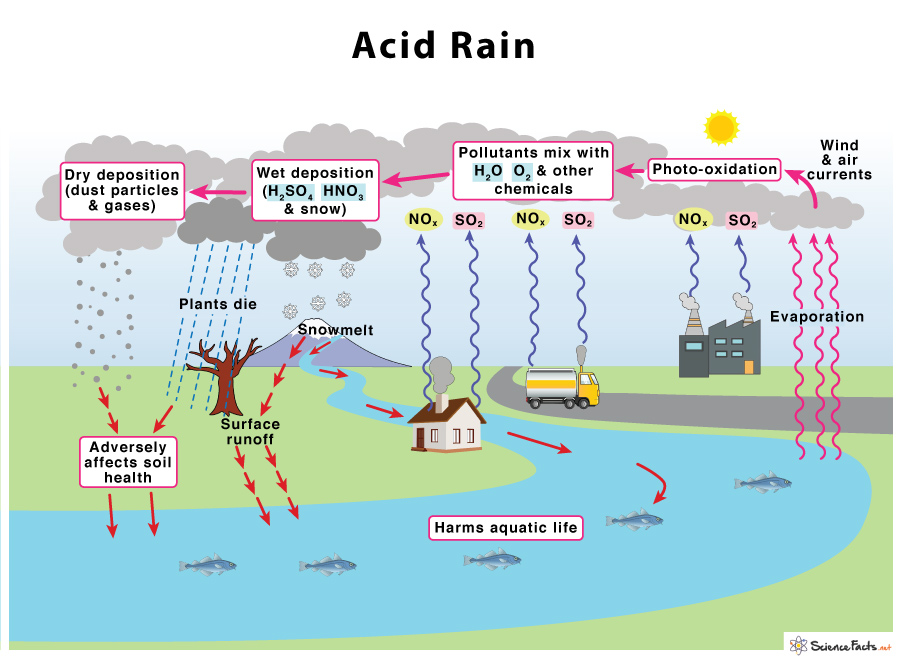
Why are the oceans and lakes getting more acidic?
Another major source of the acid that’s building up in our waters, especially the ocean, is carbon dioxide that comes from fossil fuel burning, wildfires, and the intentional fires from slash and burn deforestation. CO2 (carbon dioxide) interacts with water. It also dissolves in water and some of it forms a chemical called carbonic acid, which can dissociate when it interacts with polar water molecules, like in this picture from the Federal Ministry of Education and Research:
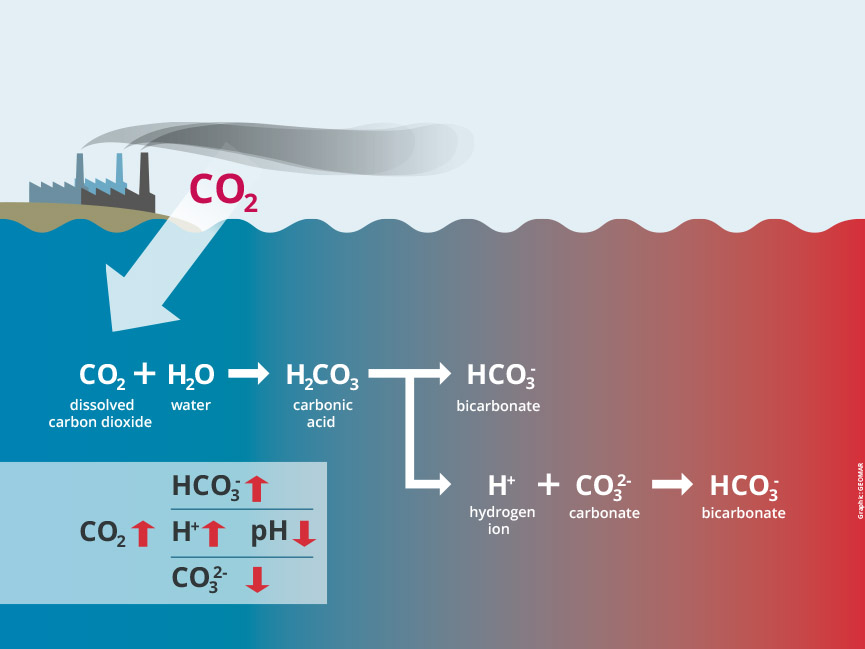
When is acid bad and why?
Acid solutions are highly reactive with other polar substances. So, acid rain can corrode iron things (rust them or even worse) and other metals. Acid rain or acidic water can damage proteins (that make up our skin, hair, nails, nasal membranes) and so cause injury or even death to animals living in acid water or soil. Acid rain can damage proteins that form plants (especially their protective waxy cuticles), so they get damaged or even die. It’s bad news. Hence the dramatic statement in the first picture, “All fish die.” The more acidic, the more reactive and thus more harmful.
Our oceans are becoming less basic, moving from pH 8.2 to about 8.1 nowadays. Some areas of the ocean have an even lower pH. This seems like such a small change, but it’s actually a big deal. It’s a 30% increase in the acid direction of the pH scale. The additional acidity reacts with the chemical that makes up sea shells and hard corals (calcium carbonate) and can dissolve it, making shells thinner. Increasing ocean temperatures also lead to shell thinning, so both changes from burning fossil fuels act together.
Coming up in the next post:
A lot of us do think about acid in our daily lives- acid reflux, acid stomach, antacids at the pharmacy. Next time, I’ll talk a little bit about pH and our physiology. Hope you’ll tune in.
This newsletter is devoted to explaining how biology works and how it is relevant to our daily lives. Most of us stopped learning about biology in high school or even middle school. And the way we learned it was as isolated concepts and vocabulary to memorize. I hope that this newsletter helps you rekindle that love of biology and might even help with better understanding some of the important biology all around us. Please share this with anyone you think might want to take a look.
Thanks for reading!




