Living in the Northeastern United States means changes in seasons. Summer to fall to winter to spring. With the warming of the planet, our seasons are less and less predictable, but one thing remains a feature of at least four months out of the year – road salt.
Anytime there’s a forecast of cold surfaces and impending precipitation, trucks filled with crystals of salt, gravel and sand drive up and down highways, town streets and parking lots, dumping out the salt mixtures, using spreaders to coat surfaces in advance of and during storms.
According to the USGS, somewhere around 30 million metric TONS of road salt are spread across the US roadways, parking lots and sidewalks every year. This is staggering!
What is road salt?
Most road salt used by municipal crews is very similar to table salt, sodium chloride. Sometimes there are additives that help with spreadability and sometimes there are also mixtures with sand or small bits of gravel. The salt helps to melt snow and ice on roadways and sidewalks by lowering the freezing point of the “saltwater.” You can try this at home: put some salty water in some ice cube tray wells and some tapwater that’s not salty and see how long each takes to freeze. There’s a big difference, which really matters during snowy/icy weather.
If the precipitation event is happening when the air temperatures are above about 15°F, road salt is pretty effective at melting snow and ice on surfaces. Below that temperature though, the salt takes longer to melt the ice and, when it gets really cold on the road surface, the salt just freezes into the ice/snow (https://canadasalt.ca/at-what-temperature-does-road-salt-stops-working/). Road salt is also less effective if the precipitation rate is too high. In both of those cases, the added sand or bits of gravel can help increase traction on the roadways. Or, road crews really pile it on, which only works when the temperatures aren’t too low.
Making mixtures of salt and grit is more expensive and time-consuming, so a lot of municipal trucks stick primarily with salt. As for the ice melts you buy for your driveway and walks at your home, you can buy calcium or magnesium-based salts to avoid the harms of sodium chloride, but they are more expensive and require more material to be effective.

Impacts of road salt on our environment
The road salt mixes with the water precipitation, forming a brine solution that runs off the roads, seeping into the soil and running into storm drains. The saltwater ends up in surface waters like ponds, lakes, streams and rivers. It also percolates through the soil into groundwater.
Victoria Kelly and Stuart Finlay from the Cary Institute of Ecosystem Studies and collaborators from Vassar College and the Cornell Cooperative Extension of Dutchess County, NY, looked at the salt concentration in well water from about 100 residential wells in the town of East Fishkill, NY and related the salt levels to the distance the well sits from a road. East Fishkill is a mostly suburban area in Dutchess County, about 60 miles north of NYC. Like most areas in the Northeast US, the roads are salted repeatedly during wintry weather events. As the salt melts snow and ice, the salty water runs off the road surfaces into the surrounding soil and also storm drains. Homeowners also dump salt on their sidewalks and driveways. This also runs off into their yards and down streets to storm drains leading to the Hudson River.
Victoria Kelly and her team found, not surprisingly, that wells closer to roads had higher salt concentrations. The salt levels were even higher if the roads are paved, as compared with gravel or dirt roads. Paved roads are impervious surfaces, and promote more runoff of liquids to a farther distance than the slower seepage that happens with a dirt or gravel road.
Here’s one of the graphs from their paper, showing that salt (measured here as chloride) levels are higher in wells closer to a road:
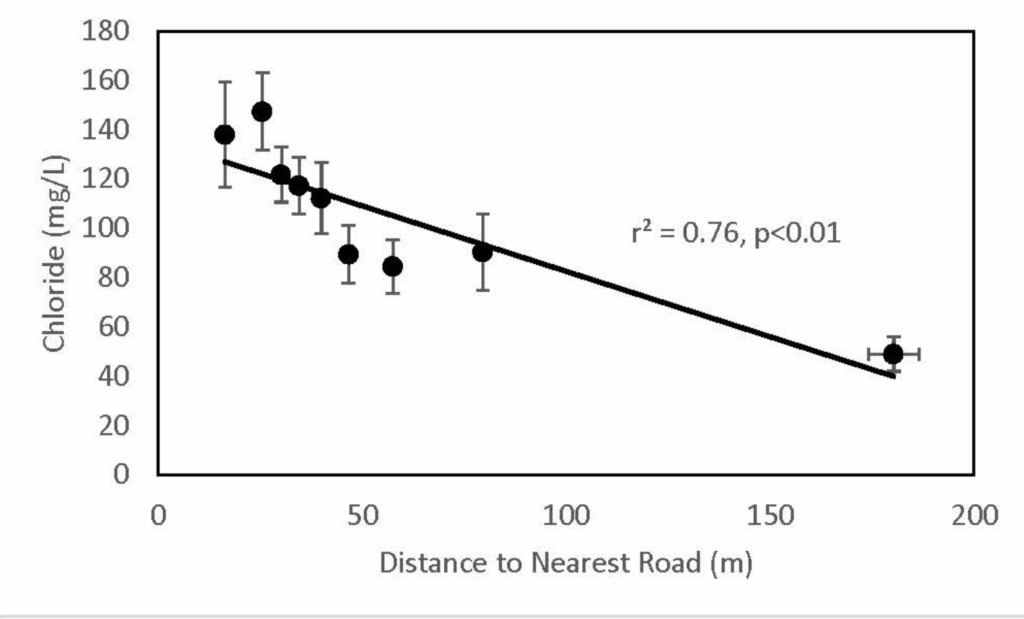
Bottom Line: Treating roads with road salt leads to saltier drinking water, particularly for well water drinkers. As we increase suburban development and create more paved roads, we are making our water supplies saltier.
Is drinking salty water harmful to us and wildlife? What’s the big deal about salty water?
Salt (especially sodium, but of course road salt, like table salt, is sodium and chloride) is required by the EPA to be at less than 20 mg/L for safe municipal drinking water. The levels found in the private, residential wells in East Fishkill in the Kelly study were as high as 860 mg/L. That’s salty! Folks on a low salt diet for hypertension or other medical conditions, are told to ingest fewer than 1500 mg of salt per day. If you drink 8 glasses (64 fluid ounces, which is almost 2 liters) of water from your well, you would ingest well over the recommended amount of salt, just from drinking water!
You might even feel more thirsty from drinking all that salt water. Too much salt intake causes higher blood pressure, which puts a burden on your heart and kidneys. Over time you’re at a high risk of heart disease, stroke, kidney disease and more.

Of course, salty water is also really bad for other organisms besides just humans. It kills plant roots, as well as harms insects, fish, birds, mammals. Overall, not good for our freshwater to be salty.
The other big thing that saltwater does is corrode things, especially metal. That’s bad for cars, but also for road structures like bridges and guardrails.


Increased salt in the soil also leaches out other harmful chemicals from the soil, which can then enter water sources. Chemicals like radium, which is a naturally occurring radioactive substance. And nitrates in the soil, which depletes soil of important sources of nitrogen for plants, but also increases nitrogen in water, which promotes harmful algae blooms.
What can we do to have safe roads in wintry conditions without so much road salt?
One thing we could consider is changing our behavior during winter weather events. Of course we need to keep roads open for emergency transportation, but do we really have to just keep chugging along, business as usual? The pandemic taught us that we can do a lot with online and remote technologies- which could mean workplaces could encourage folks to stay home and work remotely. Heck, the schools in this area close at the first snowflake, so letting folks that can work from home will reduce the number of folks on the roads. Well, one can dream.
There are ways to minimize the use of road salt. Here’s a list, compiled from a number of places:
- Pre-treat roads with a brine solution. This will reduce the initial freezing of precipation on the roads, which can reduce the overall amount of road salt used.
- Make new roads, parking lots, driveways out of porous pavement materials. This will allow the precipatation to percolate below the road, rather than sit on the surface and freeze. Porous pavements will also reduce the refreezing of puddles that happens after a storm.
- Use mixtures of salt and sand/gravel to reduce the overall use of the salt.
- Municipalities can increase the frequency of plowing, particularly early in a storm. This, combined with the brine pre-treatment, can reduce the amount of road salt.
- Be mindful of the temperatures and switch to sand/gravel rather than salt when temperatures are too low for the salt to be effective.
- Plant native plants that can tolerate higher salt levels at roadsides and in parking lots, rather than grass. This can create a buffer zone to capture the saltwater run off.
This newsletter is devoted to explaining how biology works and how it is relevant to our daily lives. Most of us stopped learning about biology in high school or even middle school. And the way we learned it was as isolated concepts and vocabulary to memorize. I hope that this newsletter helps you rekindle that love of biology and might even help with better understanding of some of the important biology all around us. Please share this with anyone you think might want to take a look.
Thanks for reading!