We are internal combustion engines. We take in carbon-based fuels and our body metabolism burns the fuel using oxygen that we breathe in, powering our body heat, movement and all our body processes. The waste we produce is mostly carbon dioxide (CO2) and water and a few other waste chemicals that show up in our urine and poop. We breathe out the carbon dioxide (and water vapor). We are a lot more efficient than trucks and power plants burning fossilized carbon fuels!
In an evolutionarily brilliant feat, the carbon dioxide that our cells produce is the major balancer of our body pH. The chemical interactions between carbon dioxide and water that happen in our cells and blood stream form a buffer to keep our pH constant.
A buffer is a chemical interaction where the H+ of acid can be absorbed or released. In this way, it can keep pH relatively constant when acids and bases enter or leave the system.
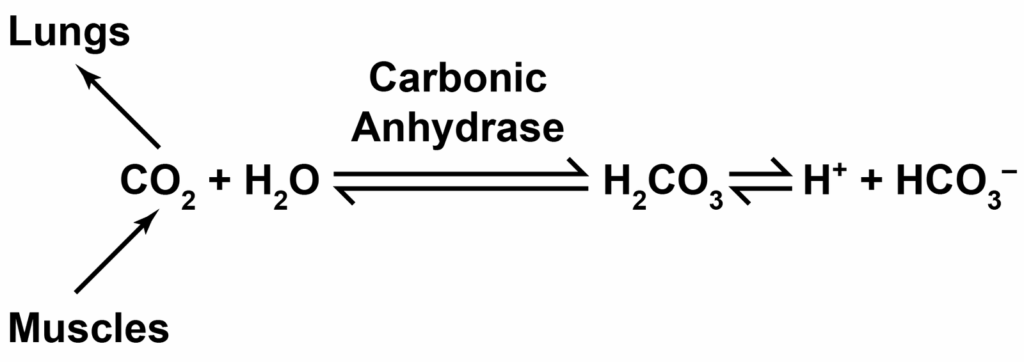
Let’s look at how CO2 forms a buffer system for our blood and body pH. You’ll maybe recognize this from an earlier newsletter on ocean acidification (it’s the same chemical system!). Our body tissues and organs, like a working muscle as an example, produces carbon dioxide and water from burning its fuel. Working muscles produce other combustion waste products, like lactic acid (a metabolic acid). The carbon dioxide and metabolic acids would acidify the blood pH without a buffer. The buffer system acts like a seesaw to keep the pH in balance. Then, lungs play a role in removing the excess carbon dioxide from all that muscle work (and the kidneys take care of the metabolic acids). Let’s dive in a little bit deeper. Really look at the equation below- it’s simple and magnificent.
You see, just like in the ocean acidification example, the carbon dioxide interacts with water and forms an acid (H2CO3) which releases H+ and forms the chemical bicarbonate (HCO3–).
The reaction is bidirectional, so if there’s more H+ around, it’ll combine with the bicarbonate, form the acid and then break apart to carbon dioxide and water. If there’s more CO2 around, the reaction moves towards producing more H+ and HCO3–. Back and forth like a seesaw.
This bidirectionality gives the chemical reaction the properties of a buffer. It can absorb H+ and go towards carbon dioxide. Or, it can release H+ (if carbon dioxide builds up, for example).
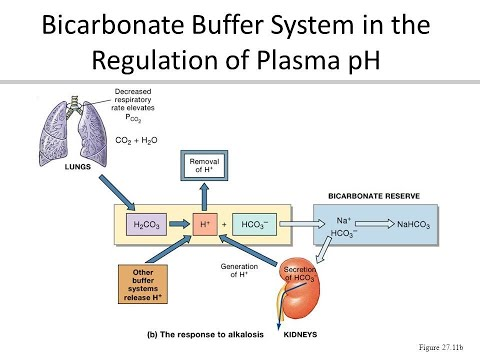
Image from: https://www.youtube.com/watch?v=pmItde9_vmw (which is a short video).
In looking at the picture above, you see what happens when you hold your breath. The CO2 levels in the blood increase, which pushes the chemical interaction towards increased H+ and HCO3–. The kidneys can work on the HCO3– levels to combine with the H+ and keep the pH balanced (although this is a slower process, so doesn’t really kick in with every breath, but would kick in if you have a persistent problem with breathing, like emphysema).
This terrific chemical reaction system happens all on its own, but our red blood cells have an enzyme to massively speed it up. The enzyme, called carbonic anhydrase, is one of the fastest enzymes known! When cells do their metabolism, the CO2 diffuses out into the blood and is rapidly converted to the bicarbonate and H+ in the blood cells and the bicarbonate is what travels in the bloodstream. This is how the majority of the CO2 travels in the blood until it gets to the lungs, instead of just as dissolved carbon dioxide. This is why our blood doesn’t resemble seltzer or a carbonated beverage.
In the lungs, the reaction goes rapidly in reverse and we breathe out the CO2. Our breathing rate is tied to the regulation of our blood pH, keeping it a steady 7.35 or so. Most of us breathe about 12-14 breaths in a minute. This steady rhythm is controlled by a region at the top of our spinal cord/base of our brain. We have cells that are sensitive to pH in the blood that signal us to breathe more if the pH falls (gets more acidic) or breathe less if the pH increases (gets more alkaline).
Here’s a helpful picture of how carbon dioxide is transported in the blood (as bicarbonate, HCO3–) from tissue (like a muscle) to lungs. The pic is from https://www.geeksforgeeks.org/biology/transport-of-carbon-dioxide-in-the-blood/.
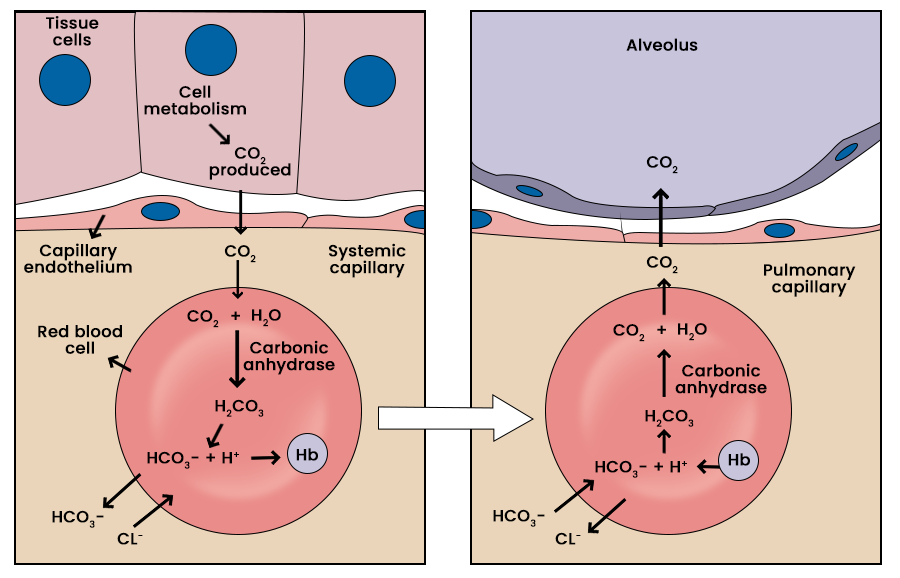
In the active tissue …………………………..> In the lung
When we exercise, our muscles speed up their metabolism, burning more fuel and producing more carbon dioxide. This will tend to push the blood pH buffer towards more H+, which will stimulate those sensors to speed up our breathing rate and depth. This draws in more oxygen, which of course is important for the metabolism, but the real goal of the sensors is to keep the pH steady, so we breathe out more carbon dioxide.
If our breathing is too slow or inadequate to match the production of carbon dioxide in our body, our blood pH can become too acidic. It’s hard to change your pH just by holding your breath (although this would work in theory, right?) because we have a very strong urge to breathe as the carbon dioxide builds up. It’s kind of fun to fiddle around with this. First take a regular breath and hold it. Time it. How long could you hold your breath?

Now, try breathing more often and more deeply for a minute (not too long or you will get dizzy). Take maybe 20 breaths instead of 14. What this does is reduce the amount of CO2 just a bit. Then hold your breath and time it. Did you hold your breath longer?
Sometime when you are exercising, try holding your breath. You’ll find you cannot hold it as long as when you’re just sitting still at rest. This is because the CO2 from your active muscles shifts the buffer towards H+, which stimulates you very strongly to breathe. CO2 itself can stimulate you to breathe, but pH is a very strong stimulus.
Kidneys help with longer term pH balance
Our kidneys take care of the bicarbonate side of that nifty blood/cell buffer system. Our kidneys filter the blood and can reabsorb more or less of the bicarbonate depending on the overall pH of the blood. If the blood pH gets out of range of this buffer system, there are other ways the kidneys can correct things and you pee out more acidic urine. This process of correcting pH imbalances by the kidneys can take hours and is key to longer-term pH balance, so it’s important that our lungs are a rapid-response system to respond to quick changes like during exercise.
pH balance inside the body is just as important as in the oceans, rivers and soil and I think it’s almost magical that the carbon dioxide-bicarbonate buffer system is a major pH balancer inside our bodies and outside in the world.
This newsletter is devoted to explaining how biology works and how it is relevant to our daily lives. Most of us stopped learning about biology in high school or even middle school. And the way we learned it was as isolated concepts and vocabulary to memorize. I hope that this newsletter helps you rekindle that love of biology and might even help with better understanding of some of the important biology all around us. Please share this with anyone you think might want to take a look.
Thanks for reading!